AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Aluminum oxide reactivity8/3/2023  
C3.2 How are metals with different reactivities extracted?.C3 Chemicals of the natural environment.4.2 Explain displacement reactions as redox reactions, in terms of gain or loss of electrons.Topic 4 - Extracting metals and equilibria.A more reactive metal can displace a less reactive metal from a compound.The protective oxide layer forms instantly the aluminium is exposed to the air. This allows a simple exchange reaction with the copper(II) sulfate. Scratches on the surface of the oxide layer allow chloride ions to react with aluminium, this effects the cohesiveness of the oxide layer.Aluminium + copper(II) sulfate → copper + aluminium sulfate Now aluminium is more reactive because it displaces copper.The aluminium foil appears unable to displace copper from copper(II) sulfate solution. Aluminium appears less reactive than copper.Remind students what copper looks like, so that they know what they are looking for. 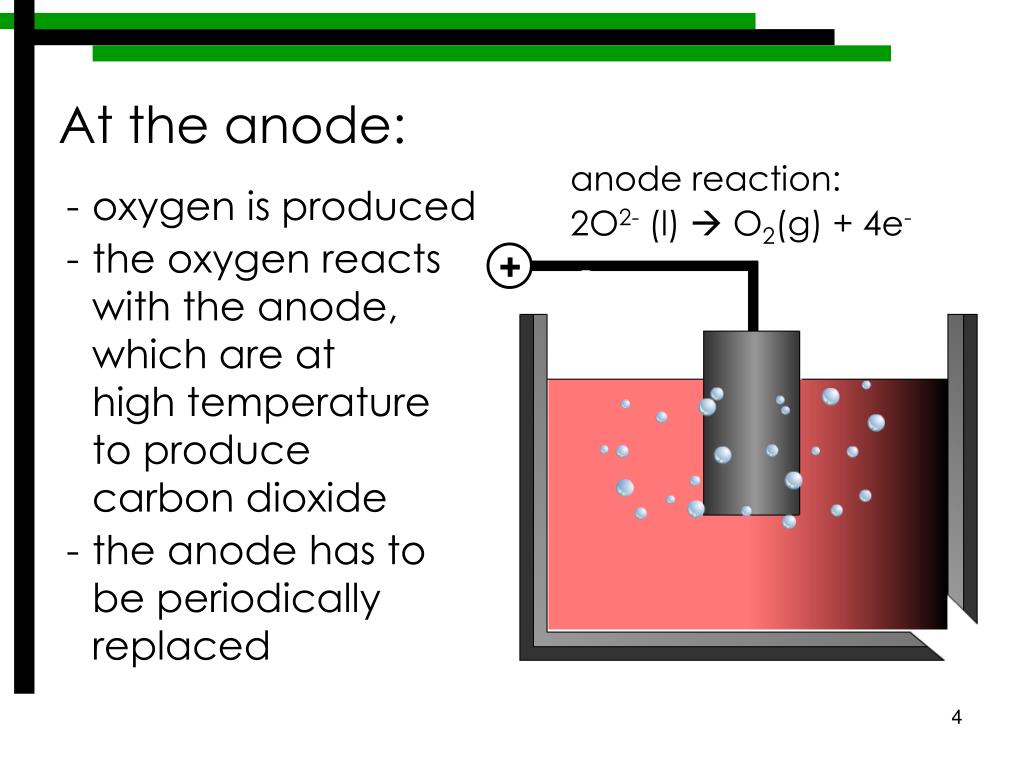
This allows reaction with the copper(II) sulfate. Scratches on the surface of the oxide layer allow chloride ions to react with aluminium, this effects the cohesiveness of the oxide layer. Sodum chloride disturbs this oxide layer.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

